In summer of 2025, I joined the research lab of Dr. Jeromy Rech at the University of North Carolina Asheville. Rech Lab aims to address the global energy crisis through developing cheap, clean, and easily synthesized solar energy.
Organic solar cells (OSCs) are a low cost, lightweight, and flexible alternative to traditional silicon-based solar panels. They are comprised of polymers that are dissolved in solvent and printed like ink to create low cost solar devices. While OSCs will likely never replace traditional silicon solar panels, they could supplement them, especially in locations that don’t work with traditional technology. Additionally, the conjugated polymers have increasingly complex synthetic routes that impede their commercial viability and have negative environmental costs.
Many polymer donors exist for use in OSCs, but Rech lab focuses on PTQ10, a polymer with an ideal balance, having a high power conversion efficiency (PCE) yet not being synthetically complex.1 The PCE of PTQ10 is over 15%, making it a viable alternative to traditional solar panels. As for its synthetic complexity, PTQ10 can be synthesized in five high-yielding steps, while other polymers can take upwards of ten steps, which adds cost and time to their production.

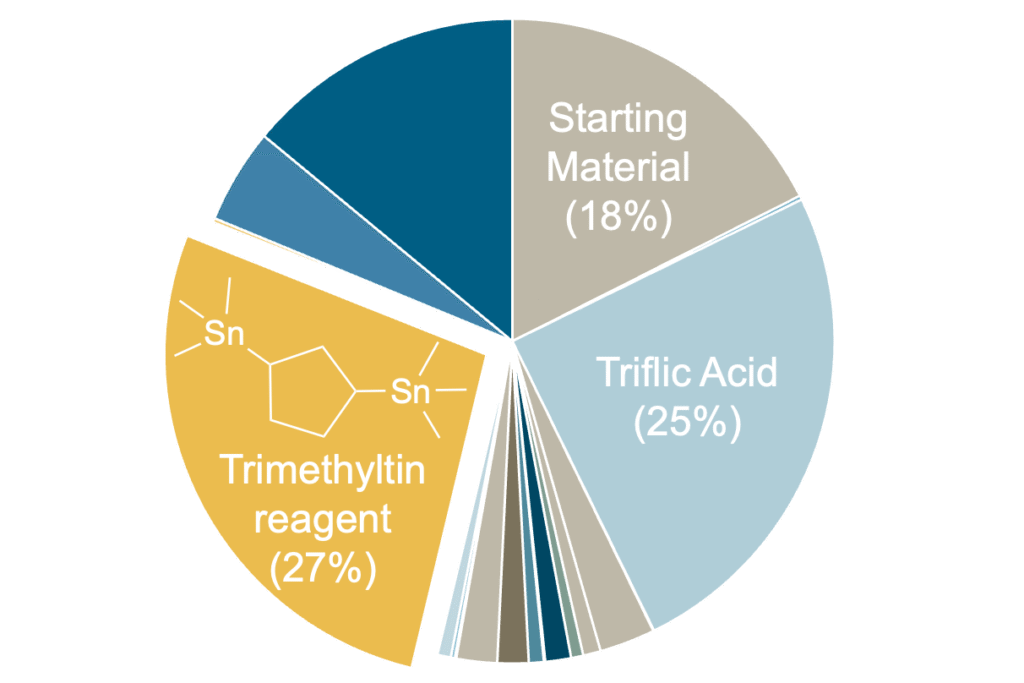
While the efficiency of PTQ10 has neared the potential to compete with traditional solar cells, cost remains a major barrier to commercial viability. Research indicates that the cost of OSCs must decrease to $0.018 Wp-1 (dollars per Watt peak) to match inorganic performance, assuming a 10% scaled-up module efficiency.2 To meet this, the organic polymer contributing to the cell must cost less than $12 g-1. Although PTQ10 has a simple synthesis pathway, total cost was calculated to be $214.18 g-1. The expense of this synthesis is due to its reliance on expensive starting materials and non-scalable reaction conditions. An updated synthesis route from Rech et al decreases the cost to $30.29 g-1 by using a Mitsunobu reaction to add the side chains and a newly redesigned and commercially available starting material. However, this work still relies on the Stille coupling for the polymerization. Over 25% of the overall cost of PTQ10 through this new synthetic approach comes from the trimethyltin chloride reagent used to make the Stille monomer. An alternative polymerization pathway could reduce the cost towards the target of $12 g-1.
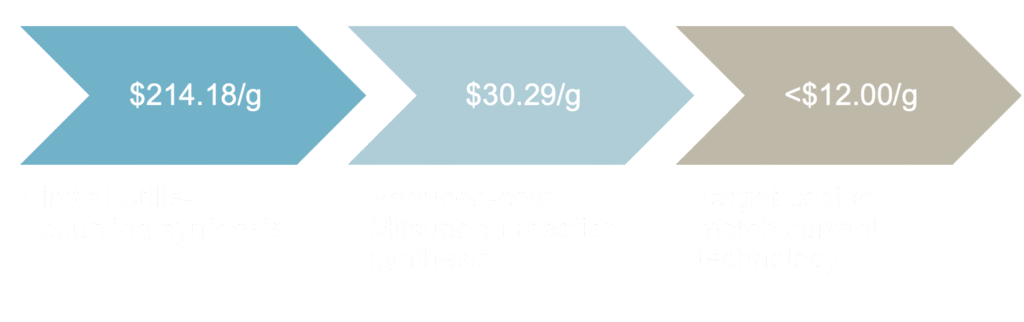
My research is aiming at a tin free polymerization methodology. We are looking to use thiazole(5,4-d)thiazole (TTz) as an acceptor unit in the polymerization of PTQ10. TTz is a highly electron-drawing unit that provides rigidity, crystallinity, and planarity to polymers, which can increase π-π stacking.3 Outside of our lab, TTz is commonly used in light-emitting diode devices and as an organic field emitting transistor.4

In current literature, TTz has often been incorporated into polymer donors for OSCs.3 However, the TTz unit is often incorporated with a traditional Stille polycondensation, meaning that this method does not suit the sustainability goals of Rech lab.
I developed three phases of research: Understanding TTz reaction conditions, functionalizing PTQ10 for use with TTz, and polymerization.

Understanding TTz
MP-1: Determining dithiooxamide reaction conditions (68% yield)


MP-2: Determining dithiooxamide reaction conditions with electron withdrawing groups (70% yield)


These experiments confirmed that the small molecule TTz synthesis was successful when reacting dithiooxamide and an aldehyde in DMF at 150 ºC for at least 8 hours. Samples were then precipitated in cold water and vacuum filtered before being recrysallized according to the literature.5 The correct molecular structure was then confirmed with 1H NMR.
MP-6: Dithiooxamide reaction conditions under microwave (32% yield)


MP-7: Dithiooxamide reaction conditions under microwave with electron withdrawing groups (<2% yield)


We tested microwave reaction conditions for the previously synthesized small molecules. Reagents were added to a microwave vial under inert conditions and irradiated under microwave at 150 ºC for 30 minutes. Samples were cooled to room temperature and dissolved in THF before being set to reflux with chloranil for 10 minutes in open air. Samples were then precipitated in cold water and recrystallized according to the literature.6,7 These were more difficult reactions to work up due to them being solvent-free reactions in the microwave–which often ended up quite burnt into the vials. More work is needed to confirm that this synthesis route can have viable yields!
MP-5: Brominating MP-1 (56% yield)


The product from MP-1 was set to reflux with NBS and DMF overnight, then vacuum filtered and rinsed with water before being recrystallized from chloroform.8
| Neat | DMF | NO2Bz | |
| 2 equivalents of aldehyde | 162% | 73% | 5% |
| 4 equivalents | 5% | 100% | 7% |
| Neat | DMF | |
| 2 equiv. | 22% | 41% |
| 4 equiv. | 5% | 50% |
Functionalization
MP-3: Converting Br to aldehyde on hexylthiophene (57% yield)

Under inert conditions at -78 ºC, hexylthiophene was reacted with n-Butyllithium and DMF overnight.9 The reaction was quenched and underwent a liquid-liquid extraction before being purified by column chromatography. This was an especially challenging reaction where we experimented with multiple purification techniques, but were unable to remove impurities.
Polymerization
To be seen!
Future concerns
- Solubility
- Chain length
Acknowledgements
References
(1) Rech, J. J.; Neu, J.; Qin, Y.; Samson, S.; Shanahan, J.; Josey III, R. F.; Ade, H.; You, W. Designing Simple Conjugated Polymers for Scalable and Efficient Organic Solar Cells. ChemSusChem 2021, 14 (17), 3561–3568. https://doi.org/10.1002/cssc.202100910.
(2) Machui, F.; Hösel, M.; Li, N.; Spyropoulos, G. D.; Ameri, T.; Søndergaard, R. R.; Jørgensen, M.; Scheel, A.; Gaiser, D.; Kreul, K.; Lenssen, D.; Legros, M.; Lemaitre, N.; Vilkman, M.; Välimäki, M.; Nordman, S.; Brabec, C. J.; Krebs, F. C. Cost Analysis of Roll-to-Roll Fabricated ITO Free Single and Tandem Organic Solar Modules Based on Data from Manufacture. Energy Environ. Sci. 2014, 7 (9), 2792–2802. https://doi.org/10.1039/C4EE01222D.
(3) Ji, M.; Dong, C.; Guo, Q.; Du, M.; Guo, Q.; Sun, X.; Wang, E.; Zhou, E. Recent Advances in Organic Photovoltaic Materials Based on Thiazole‐Containing Heterocycles. Macromol. Rapid Commun. 2023, 44 (13). https://doi.org/10.1002/marc.202300102.
(4) Woodward, A. N.; Kolesar, J. M.; Hall, S. R.; Saleh, N.-A.; Jones, D. S.; Walter, M. G. Thiazolothiazole Fluorophores Exhibiting Strong Fluorescence and Viologen-Like Reversible Electrochromism. J. Am. Chem. Soc. 2017, 139 (25), 8467–8473. https://doi.org/10.1021/jacs.7b01005.
(5) Adams, T. J.; Brotherton, A. R.; Molai, J. A.; Parmar, N.; Palmer, J. R.; Sandor, K. A.; Walter, M. G. Obtaining Reversible, High Contrast Electrochromism, Electrofluorochromism, and Photochromism in an Aqueous Hydrogel Device Using Chromogenic Thiazolothiazoles. Adv. Funct. Mater. 2021, 31 (36), 2103408. https://doi.org/10.1002/adfm.202103408.
(6) Dessì, A.; Calamante, M.; Mordini, A.; Zani, L.; Taddei, M.; Reginato, G. Microwave-Activated Synthesis of Thiazolo[5,4-d]Thiazoles by a Condensation/Oxidation Sequence. RSC Adv 2014, 4 (3), 1322–1328. https://doi.org/10.1039/C3RA45015E.
(7) Papernaya, L. K.; Shatrova, A. A.; Sterkhova, I. V.; Levkovskaya, G. G.; Rozentsveig, I. B. Microwave-Assisted Synthesis of 2,5-Diarylthiazolo[5,4-d]Thiazoles from Benzaldehydes and Dithiooxamide. Russ. J. Org. Chem. 2015, 51 (3), 373–377. https://doi.org/10.1134/S1070428015030148.
(8) Jung, I. H.; Yu, J.; Jeong, E.; Kim, J.; Kwon, S.; Kong, H.; Lee, K.; Woo, H. Y.; Shim, H.-K. Synthesis and Photovoltaic Properties of Cyclopentadithiophene-Based Low-Bandgap Copolymers That Contain Electron-Withdrawing Thiazole Derivatives. Chem. – Eur. J. 2010, 16 (12), 3743–3752. https://doi.org/10.1002/chem.200903064.
(9) Seo, J. H.; Nam, S. Y.; Lee, K.-S.; Kim, T.-D.; Cho, S. The Effect of Processing Additive on Aggregated Fullerene Derivatives in Bulk-Heterojunction Polymer Solar Cells. Org. Electron. 2012, 13 (4), 570–578. https://doi.org/10.1016/j.orgel.2011.12.020.
